|
POSS - Introduction
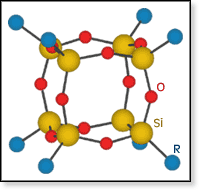 The general problem of enhancing properties of materials to increase physical and chemical durability is the focus of much current research. One strategy is to incorporate inorganic components into existing organic polymer systems. Polyhedral oligomeric silsesquioxane (POSS) cages are promising candidates for this approach. POSS consists of an inorganic silicon-oxygen cage with the general composition SiO3/2 and organic substituents R capping the silicon atoms of the cage. The overall formula is normally written RnTn, where T = SiO3/2. The figure at right schematically shows the structure for n = 8, but cages with n = 6, 10, 12, 14 and 16 also exist. The general problem of enhancing properties of materials to increase physical and chemical durability is the focus of much current research. One strategy is to incorporate inorganic components into existing organic polymer systems. Polyhedral oligomeric silsesquioxane (POSS) cages are promising candidates for this approach. POSS consists of an inorganic silicon-oxygen cage with the general composition SiO3/2 and organic substituents R capping the silicon atoms of the cage. The overall formula is normally written RnTn, where T = SiO3/2. The figure at right schematically shows the structure for n = 8, but cages with n = 6, 10, 12, 14 and 16 also exist.
Common POSS R groups include Me, Et, iPr, iBu, iOc (isooctyl), Ph, c-C5H9, c-C6H11, and c-C7H13. Substituting one or more of these R groups with a reactive functional group allows POSS cages to be incorporated into organic polymer systems. The resulting hybrids have characteristics between those of traditional organic systems (polymers) and traditional inorganic systems (ceramics). The goal is to produce materials that combine the advantages of standard polymers, such as processability, toughness and low cost, with the advantages of ceramics, such as thermal and oxidative stability. Exactly how the addition of POSS to organic polymers brings about these improvements is unknown. One of the major issues receiving attention is how the POSS units integrate with the polymer as a whole. It usually is not known how they are distributed nor how the polymer conformation adapts to the presence of the POSS cages. What has been observed in a number of systems is that the POSS cages tend to aggregate forming POSS-rich domains within a polymer matrix.
In order to better design POSS-containing polymers, the relationship between molecular structure and the physical properties of the materials must be understood. By using ion mobility mass spectrometry in conjunction with extensive molecular modeling we are making progress towards that goal. We’ve begun this project by examining POSS monomers to test how our methods work with these materials and examine the effects of different R groups. We've also looked at siloxane oligomers of POSS. These are not POSS groups incorporated into a standard organic polymer but POSS cages directly linked to one another by a single oxygen atom. We’ve also begun work on hybrid POSS-polymer materials by looking at POSS cages attached to poly(propyl methacrylate). We hope to expand our work to other POSS-polymers in the future.
Projects:
References:
- "Application of Ion Mobility to the Gas-Phase Conformational Analysis of Polyhedral Oligomeric Silsesquioxanes (POSS)" J. Gidden, P. R. Kemper, E. Shammel, D. P. Fee, S. Anderson, M. T. Bowers Int. J. Mass Spectrom. 2003, 222, 63-73
- "3-Dimensional Structural Characterization of Cationized Polyhedral Oligomeric Silsesquioxanes (POSS) with Styryl and Phenylethyl Capping Agents" E. S. Baker, J. Gidden, D. P. Fee, P. R. Kemper, S. E. Anderson, M. T. Bowers Int. J. Mass Spectrom. 2003, 227, 205-216
- "Isomeric Structural Characterization of Polyhedral Oligomeric Silsesquioxanes (POSS) with Styryl and Epoxy Phenyl Capping Agents" E. S. Baker, J. Gidden, S. E. Anderson, T. S. Haddad, M. T. Bowers Nano Lett. 2004, 4, 779-785
- "Structural Characterization of POSS Siloxane Dimer and Trimer" S. E. Anderson, C. Mitchell, T. S. Haddad, A. Vij, J. J. Schwab, M. T. Bowers Chem. Mater. 2006, 18, 1490-1497
- "Structure of Hybrid Polyhedral Oligomeric Silsesquioxane Propyl Methacrylate Oligomers Using Ion Mobility Mass Spectrometry and Molecular Mechanics" S. E. Anderson, E. S. Baker, C. Mitchell, T. S. Haddad, M. T. Bowers Chem. Mater. 2005, 17, 2537-2545
|


