|
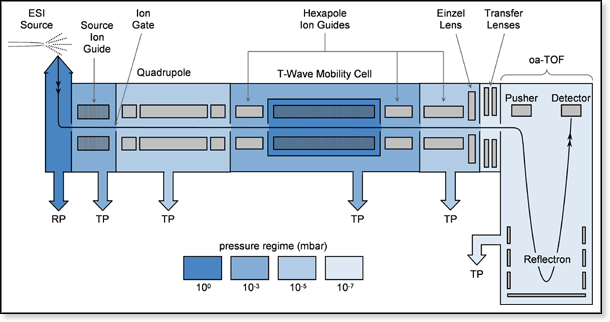
This mass spectrometer is the prototype of a commercial instrument built by Waters Corp. (Milford, MA, USA). The details of the prototype design are given in Giles, K.; Pringle, S. D.; Worthington, K. R.; Little, D.; Wildgoose, J. L.; Bateman, R. H. Rapid Commun. Mass Spectrom. 2004, 18, 2401-2414, while the new commercial instrument is described in Pringle, S. D.; Giles, K.; Wildgoose, J. L.; Williams, J. P.; Slade, S. E.; Thalassinos, K; Bateman, R. H.; Bowers, M. T.; Scrivens, J. H. Int. J. Mass Spectrom. in press, available online Sept. 6, 2006. The design of the prototype, now at UCSB, is based on Waters quadrupole time-of-flight (Q-TOF) model, but has been modified to include a mobility cell. The instrument is shown schematically below. Ions are formed by ESI in the source and sent through a quadrupole for mass selection before injection into the mobility cell. Unlike our other instruments which use a uniform electric field across the cell for ion mobility experiments, this one uses traveling wave (T-wave) technology. The T-wave cell consists of a stacked-ring RF ion guide which incorporates a repeating sequence of transient voltages applied to the ring electrodes. These voltage pulses set up a traveling electric field that propels ions through the background gas present in the mobility cell. The time it takes for an ion to drift through the cell depends on its mobility as well as the wave period and height and the gas pressure. Ions with high mobility are better able to keep up with traveling waves and are pushed more quickly through the cell. Ions with low mobility crest over the waves more often and have to wait for subsequent waves to push them forward, resulting in longer drift times. Details of the T-wave mobility method can be found in the papers cited above. To measure an arrival time distribution (ATD), ions are pulsed into the mobility cell. This triggers a series of orthogonal acceleration voltage pulses that send ions exiting the mobility cell into the TOF mass analyzer and finally to the detector. The sum of all detected ions is the ion chromatograph. Selecting a peak in the chromatograph displays the underlying TOF mass spectrum.
The Bowers group received this instrument in November of 2006 and we are just beginning to explore its capabilities. The use of T-wave technology in ion mobility mass spectrometry results in an instrument with high sensitivity and rapid data acquisition capabilities. In contrast to our other ion mobility instruments which have drift cells with uniform electric fields, the T-wave instrument cannot be used for direct measurement of collision cross sections - at least for now. As the T-wave technology matures, however, we expect that a calibration procedure will be developed. In fact this has already begun with Carol Robinson's group at Cambridge comparing the drift times of a number of proteins in a T-wave instrument to cross sections measured by traditional mobility methods (Ruotolo, B. T.; Giles, K.; Campuzano, I.; Sandercock, A. M.; Bateman, R. H.; Robinson, C. V. Science 2005, 310, 1658-1661). Our group has also done some calibration of the T-wave method using oligosaccharides (manuscript in preparation).
|